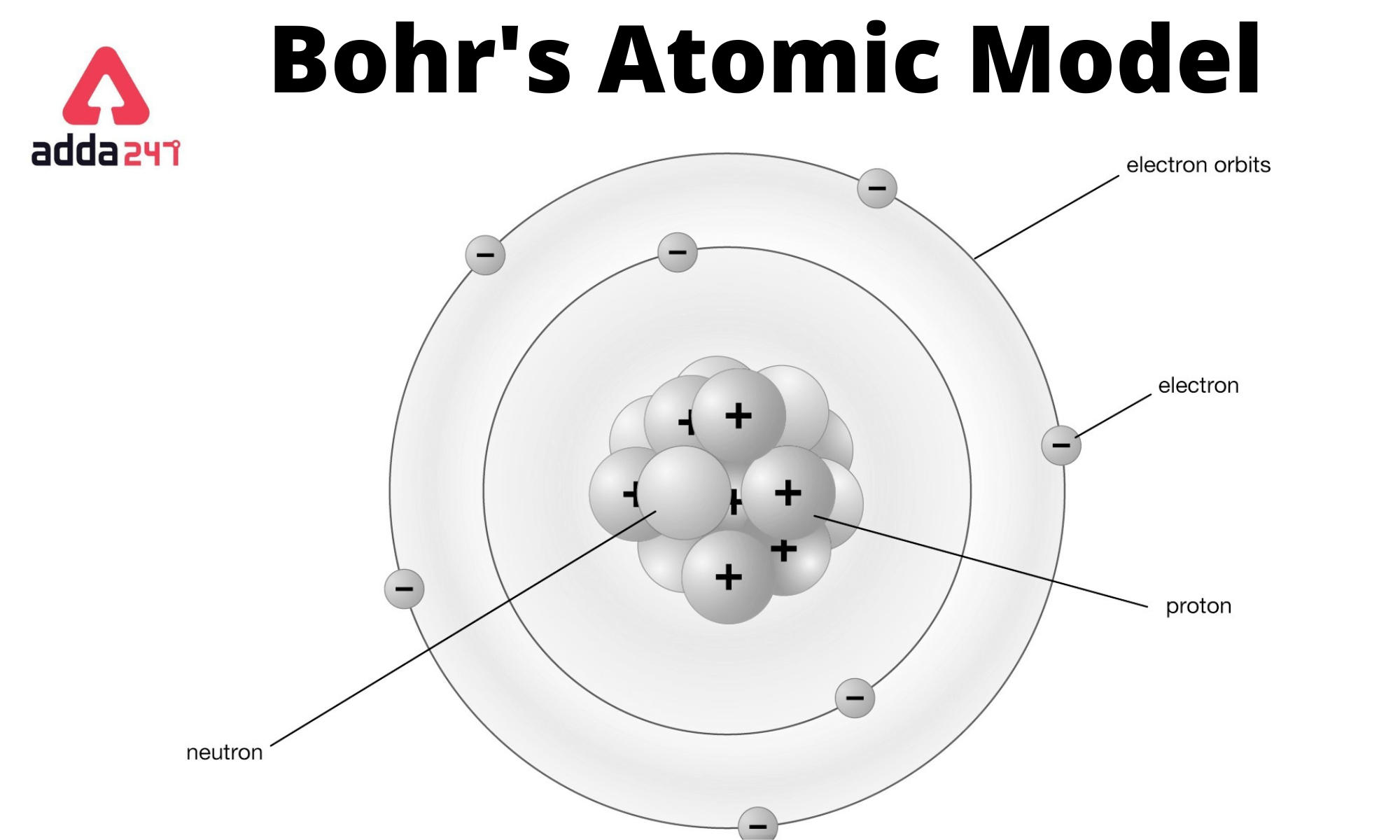
In the late 1800s, the cathode ray tube was developed and used in several investigations. The discovery of the first subatomic particle, the electron, resulted from experiments involving the effects of electricity on matter. Advances in modern physics allowed scientists to find a large number of subatomic particles, including the quarks that are the fundamental subatomic particles that form protons and neutrons. The discovery of these subatomic particles, along with Dalton ’s atomic theory, set in motion the development of several atomic models. The number of protons determines the chemical identity of the element. Atoms of the same element can differ in the number of neutrons (called isotopes) and also in the number of electrons (called ions). The number and arrangement of each of these particles within the atom determine the chemical properties of the element. The protons and neutrons are located in a central region of the atom called the nucleus, and the electrons occupy the space surrounding the nucleus. In the late 1800s, scientists discovered that atoms are composed of three subatomic particles, called protons, neutrons, and electrons. The evolution of the atomic model has led to the discovery that atoms are divisible into smaller particles, and that not all atoms of a given element are exactly the same. Not all of Dalton ’s atomic theory has stood the test of time. These five postulates not only explained the laws of conservation of mass, definite proportions, and multiple proportions, but also served as the basis for the study and development of various atomic models. Lastly, chemical reactions involve the combination, separation, and rearrangement of atoms. Fourth, atoms of different elements combine to form compounds. Third, atoms cannot be further divided, created, nor destroyed. Second, the atoms of a given element are all exactly the same in structure and properties. The first postulate states that all matter is composed of atoms. In 1808, John Dalton (1766 –1844) proposed his atomic theory, which served as an explanation for these phenomena. The law of conservation of mass, the law of definite proportions, and the law of multiple proportions provided models for the behavior of chemical reactions, but the laws could not be explained. Early atomic theoryĪtomic models have their beginnings in the early atomic theory. His ideas were met with much criticism, and it was not until the early 1800s that the atomic theory took complete hold in scientific thought. Around 450 BC, the Greek philosopher Democritus proposed that all matter is made up of small, indivisible particles called atomos (meaning indivisible). The existence of atoms was first guessed as early as 400 BC, when Greek philosophers debated whether one could divide a substance into infinitely smaller pieces or if eventually a smallest, indivisible particle would be reached. The atom is defined as the smallest part of an element that retains the chemical properties of that element.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
